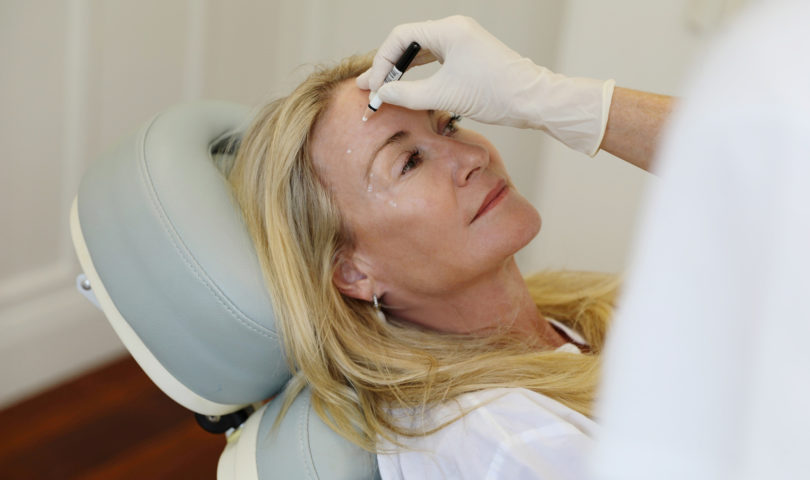
For many of us, a well-rounded skincare routine can be one that includes cosmetic procedures like injectables. Often utilised to give the impression of a more youthful appearance, what previously may have been an area of beauty that was kept under wraps has never been more open — and accepted.
Globally, the demand for fillers and injectables is on the rise, with the International Society of Aesthetic Plastic Surgery reporting an 8.6 percent increase in non-surgical injectables in 2019. The most commonly used of these is, unsurprisingly, botulinum toxin, with the society reporting 6,271,488 procedures carried out across over 15 countries including the United States, the United Kingdom, Brazil, Russia and Japan (this includes both women and men).
While these statistics for the last couple of years will undoubtedly have been disrupted by the Covid-19 pandemic, anecdotally the demand for non-surgical treatments like anti-wrinkle injectables is widely reported to be thriving. Some interesting and previously unexpected factors have influenced the conversation.
Many of us are spending more time on Zoom and FaceTime than ever before and as a possible result, we may have grown more accustomed than we’d probably like to all the nuances of our faces, including those we’re not too fond of.
When it comes to the formulation of botulinum toxin products, all of these are effective, but there are differences on how they are made and their protein content. What many people might not know, is that resistance can form against these products, through the development of antibodies against the toxin proteins. Increasing therapeutic evidence and published clinical research show that some of the risk factors which influence the build up of resistance to botulinum toxins are high doses, frequency of treatment and level of protein injected.
This resistance may result in clients requiring larger doses to keep wrinkles at bay and having to get injections more regularly. Quite apart from aesthetic concerns, botulinum toxin resistance can impact potential therapeutic treatment for conditions including musculoskeletal disorders, blepharospasm or spasticity after a stroke, as resistance can last for a significant period of time.
Aside from taking a lengthy break from injections, there is no immediate cure for resistance. Using the minimum effective dose, maximising the time between treatments and minimising the level of protein injected can all help minimise the risk of resistance.
In New Zealand, a product from leading global company Merz Aesthetics has proven effective in numerous clinical studies. Named XEOMIN®, this purified* botulinum toxin product goes through additional purification steps in the manufacturing process to remove the complexing proteins, meaning the end product contains only the active part of the botulinum toxin.
In treatment for severe glabellar frown lines, over 95 percent of patients responded by day 30, with the majority of patients maintaining a desirable effect up to four months on. Another study has also shown that XEOMIN® can help patients appear to others as looking younger and healthier.
Most of us are now, more than ever, aware of every little thing we put into (and on) our bodies — from food and drink to cosmetics, skincare and more. If this is something you put particular emphasis on, and are considering an anti-wrinkle injectable, ask your healthcare professional to see if XEOMIN® is right for you.
* XEOMIN® contains zero complexing proteins.
Xeomin® (Incobotulinumtoxin A) 50, 100 Units is a Prescription Medicine. Indications: In adults, for the treatment of cervical dystonia; blepharospasm; spasticity of the upper limb; upper facial lines: glabellar frown lines, lateral periorbital lines (crow’s feet), horizontal forehead lines. Xeomin® has both risks and benefits, consult your doctor if Xeomin® is right for you. Further information on the risks and benefits of Xeomin® can be found in the Consumer Medicine Information (CMI) available from www.medsafe.govt.nz or by calling 0800 822 310. Use strictly as directed. If symptoms continue or you have side effects, see your doctor, pharmacist or health care professional. Common side effects include: Headaches; nausea; tenderness, swelling, redness, numbness or bruising of the skin; dry eye; heavy feeling of eyelid/eyebrow/forehead; face/brow not symmetrical, drooping eyelids/eyebrows. Serious side effects are rare and include allergic reactions. Xeomin® is an unfunded medicine, prescription charge will apply. Normal doctor charges will still apply. Copyright ©2022. Pharmacy Retailing NZ Limited t/a Health Care Logistics (HCL) 58 Richard Pearse Drive, Mangere, Auckland 2022. All rights reserved. Xeomin® and Merz Aesthetics are registered trademarks of Merz Pharma GMbH & Co. KGaA. NZ_XEO_ADV_DEN_V1:MAR22 Date of preparation: March 2022. TAPS MR7602. For information about how Merz handles personal data, please see Merz General Data Protection Information Notice at www.merz.com/fin. Guideline for DTCA. Prescription medicines.
For more information on XEOMIN® and how to find your local clinic, visit –